|
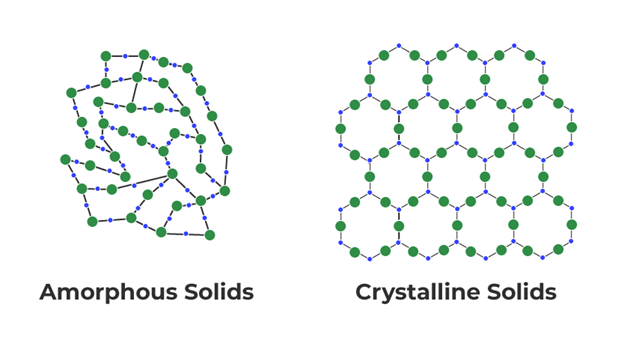
By Gabrielle Born There are 2 types of solids: Crystalline and amorphous. Crystalline solids have fixed, uniform edges and sharp melting points, while amorphous solids have curved or nonuniform edges and have a wide melting point range. Amorphous ice is an amorphous solid form of water. Where can amorphous ice be found?
There are different densities of amorphous ice. Medium-density amorphous ice, which appears like a fine white powder, could exist inside the ice moons of the outer solar system. Low-density amorphous ice is usually formed in laboratories by water vapor molecules accumulating onto a smooth, metal, crystal surface under -244°F. Amorphous ice is mostly found in outer space. It is found especially on icy moons (moons consisting mostly of ice). In outer space it is expected to be formed in a similar manner on a variety of cold substrates, such as dust particles High density amorphous ice (HDA) can be formed by compressing ice Ih at temperatures below ~140 K. How is amorphous ice different from regular ice? Regular ice is made from crystalline matter. Amorphous ice is made from amorphous matter. Common ice is a crystalline material wherein the molecules are regularly arranged in a hexagonal lattice, whereas amorphous ice lacks long-range order in its molecular arrangement. Is it okay to consume? Yes, it is. Amorphous ice is okay to consume because it is water--H2O. Since H2O is edible, then amorphous ice is okay to consume.
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
StaffLearn more about the people behind the stories by clicking the button below. Archives
October 2023
Categories |

 RSS Feed
RSS Feed
